Is ORS hypotonic or isotonic?

Sophia Vance
Published Jan 09, 2026
A hypotonic oral rehydration salts (ORS) solution with total osmolality of 224 mosmol/l was compared in an open clinical trial with an isotonic (osmolality 304 mosmol/l) ORS solution for the treatment of dehydration due to acute diarrhoea.
What type of solution is ORS?
Oral rehydration solution (ORS) is one liter of water, six teaspoons of sugar, and a half teaspoon of salt. This simple mixture has saved millions of lives and costs just pennies. ORS is the cornerstone of diarrhea treatment in low-resource settings.What is the tonicity of ORS?
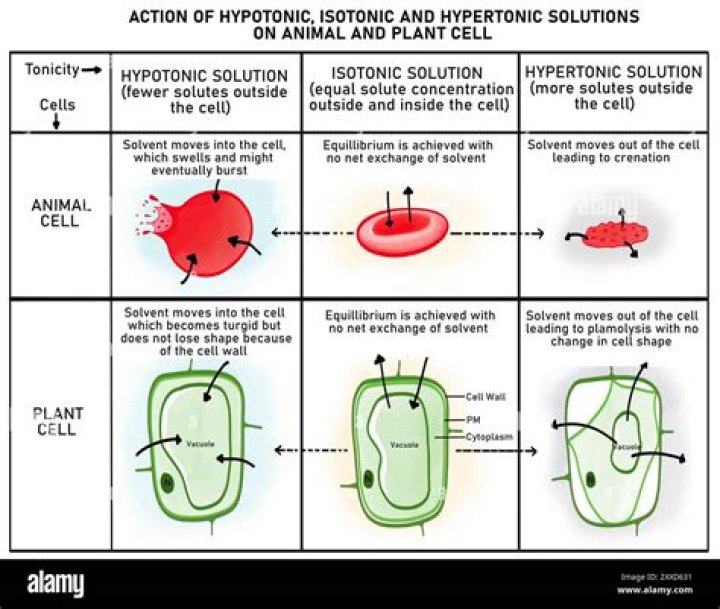
Fluid tonicity had a major effect on the uptake and elimination of the ORS. The hypertonic fluid (628 mOsm/kg bwt) was less rapidly absorbed and resulted in more rapid fluid and electrolyte excretion than the isotonic (314 mOsm/kg bwt) and hypotonic (water) fluids.WHO ORS osmolarity?
For more than two decades, the World Health Organization (WHO) has recommended the standard formulation of glucose‐based ORS with 90 mmol/L of sodium and 111 mmol/L of glucose and a total osmolarity of 311 mmol/L.What is the mode of action of ORS?
The efficacy of ORS is based on the ability of glucose to stimulate Na and fluid absorption in the small intestine via a cyclic AMP-independent process.Hypertonic, Hypotonic and Isotonic Solutions!
Is oral rehydration salt hypertonic?
rehydration solutions are mildly or moderately hypertonic. rehydration solutions is by clinical trial.Are oral rehydration solutions hypotonic?
A hypotonic oral rehydration salts (ORS) solution with total osmolality of 224 mosmol/l was compared in an open clinical trial with an isotonic (osmolality 304 mosmol/l) ORS solution for the treatment of dehydration due to acute diarrhoea.Why does ORS have a low osmolarity?
Why “reduced osmolarity”? Studies have shown that the efficacy of ORS for treatment of children with acute diarrhoea is improved by reducing its sodium concentration to 75 mEq/l, its glucose concentration to 75 mmol/l, and its total osmolarity to 245 mOsm/l.WHO ORS vs low osmolarity ORS?
However, patients treated with reduced-osmolarity ORS solution had a significantly lower volume of vomiting and significantly higher urine output than those treated with standard WHO-ORS solution. Reduced-osmolarity ORS solution was as efficacious as standard WHO-ORS solution in the management of cholera patients.Why is osmolality reduced in ORS?
One successful approach is based on reducing the osmolarity of ORS solution to avoid possible adverse effects of hypertonicity on net fluid absorption. This was done by reducing the solution's glucose and salt (NaCl) concentrations.Is Hydralyte hypotonic?
Hydralyte Sports, however, is a hypotonic Oral Rehydration Solution (ORS), which means it has a lower concentration of solutes than your cells.What causes hypotonic dehydration?
Hypotonic dehydration occurs when sodium loss is greater than water loss, resulting in a decrease in serum osmolality. This causes a shift of water from the extracellular space into the intracellular space. The cells swell and cerebral edema may occur. Hyponatremia can be acute or chronic.What is ORS and its composition?
Oral rehydration solution (ORS) is an oral powder–containing mixture of glucose sodium chloride, potassium chloride, and sodium citrate. After being dissolved in the requisite volume of water they are intended for the prevention and treatment of dehydration due to diarrhea, including maintenance therapy.Is ORS same as normal saline?
No. They are not the same thing at all. The "complete formula" ORS contains potassium and a base - either bicarbonate or citrate - which corrects acidosis which small infants in particular suffer when they are dehydrated.What are the types of ORS?
World ORS Day 2020: 5 Different Types Of Life-Saving Oral Rehydration Salts And Their Benefits
- Advantages Of ORS.
- Low Osmolarity ORS:
- Tri-Sodium Citrate ORS:
- Rice-Based ORS:
- Home Based ORS.
- Super ORS: