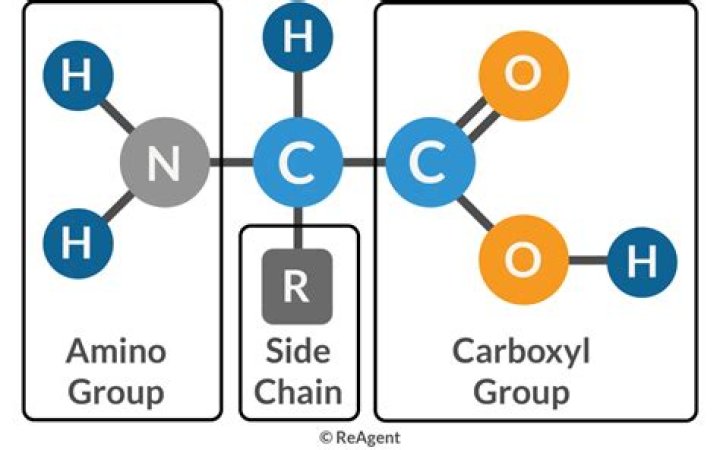
Which option explains why this amino acid can act as both an acid and a base?

Andrew Rivera
Published Jan 11, 2026
Amino acids can act as both an acid and a base due to the presence of the amino and carboxyl functional groups. The pH at which a given amino acid exists in solution as a zwitterion is called the isoelectric point (pI).
zwitterion
In chemistry, a zwitterion (/ˈtsvɪtəˌraɪən/ TSVIT-ə-rye-ən; from German Zwitter [ˈtsvɪtɐ] 'hermaphrodite'), also called an inner salt or dipolar ion, is a molecule that contains an equal number of positively- and negatively-charged functional groups.
› wiki › Zwitterion
Which amino acid can act as both acid and base?
Histidine, lysine, and arginine are considered basic amino acids because each of their side chains has a nitrogen-containing group that can exist in either a protonated or deprotonated form. However, histidine has a pKa in the acidic range.Why does the amino group act as an base?
According to the Khan Academy, the amino functional group is basic because it can remove H+ from systems.What are acidic and basic amino acids?
Compounds such as amino acids that can act as either an acid or a base are called amphoteric. The basic amino group typically has a pKa between 9 and 10, while the acidic α-carboxyl group has a pKa that is usually close to 2 (a very low value for carboxyls).How amino acids react with acids and bases?
Amino acids react with each other in a typical acid-base neutralization reaction to form a salt. The reaction is simply the transfer of the -H (positive ion) from the acid to the amine and the attraction of the positive and negative charges.Amino acids as acids and bases
What is the process in which the basic group of one amino acid and the acidic group of the other are both made unable to react?
What is the process in which the basic group of one amino acid and the acidic group of the other are both made unable to react? Clarification: The process in which the basic group of one amino acid and the acidic group of the other are both made unable to react is called protection.Which term describes a substance that could react as an acid and base?
These compounds, such as Al2O3 and Al(OH)3, are called amphoteric (literally, "either or both") because they can act as either acids or bases. Al(OH)3, for example, acts as an acid when it reacts with a base.What is meant by basic amino acid?
Basic Amino Acids. Basic amino acids are polar and positively charged at pH values below their pKa's, and are very hydrophilic.Which of the following pairs of amino acids can form hydrogen bonds between their side chain groups?
3 amino acids (arginine, lysine and tryptophan) have hydrogen donor atoms in their side chains.Which group on an amino acid acts as a base?
Zwitterions in simple amino acid solutionsAn amino acid has both a basic amine group and an acidic carboxylic acid group. There is an internal transfer of a hydrogen ion from the -COOH group to the -NH2 group to leave an ion with both a negative charge and a positive charge. This is called a zwitterion.