Why does DNA dissolve in water?

David Mccullough
Published Jan 20, 2026
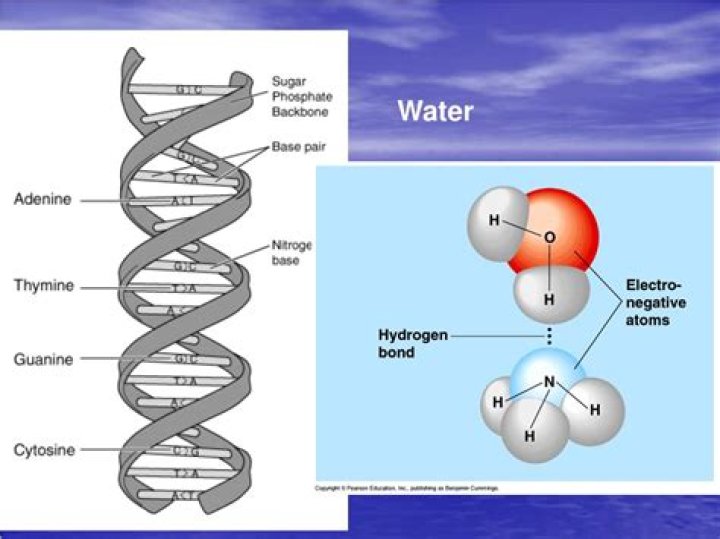
DNA is polar due to its highly charged phosphate backbone. Its polarity makes it water-soluble (water is polar) according to the principle "like dissolves like".
What happens when you dissolve DNA in water?
DNA is soluble in water. That means it can dissolve in water. However, it is not soluble when alcohol and salt are present. Lab technicians can add ethanol or isopropyl alcohol (rubbing alcohol) so that the DNA clumps and form a visible white precipitate.Why does DNA dissolve in water but not alcohol?
Because of these charges, polar molecules like DNA or RNA can interact electrostatically with the water molecules, allowing them to easily dissolve in water. Polar molecules can therefore be described as hydrophilic and non-polar molecules, which can't easily interact with water molecules, are hydrophobic.What properties of DNA makes it soluble in water?
Because DNA is usually soluble in aqueous mixtures, due to the phosphate-sugar-phosphate bonds in the backbone of the DNA, in order to isolate the DNA you must reduce its solubility in water.Does DNA precipitate in water?
It's possible for DNA to bind with water but because of the “lower dielectric constant alcohol”, the complex of DNA and salt is protected by an alcohol shield. And henceforth it can't react with water. This chemical reaction makes it possible to visualize DNA like a cotton thread and aggregates as a precipitate.Is it Dangerous To Eat Pure DNA? Extracting and Eating Pure DNA
Is the DNA soluble in the aqueous solution or alcohol?
Answer and Explanation: DNA is soluble in an aqueous solution. DNA is a polar molecule due to its sugar-phosphate backbone.Why does DNA float alcohol?
Alcohol is less dense than water, so it floats on top. Look for clumps of white stringy stuff where the water and alcohol layers meet. DNA precipitates when in the presence of alcohol, which means it doesn't dissolve in alcohol. This causes the DNA to clump together when there is a lot of it.Is DNA soluble in water and if so why?
These molecules are also polar because of the negatively charged phosphate group (PO3-) along the sugar-phosophate backbone. Because of this, DNA and RNA can easily dissolve in water.What part of DNA makes it soluble in water?
DNA is polar due to its highly charged phosphate backbone. Its polarity makes it water-soluble (water is polar) according to the principle "like dissolves like".Why do polar molecules dissolve in water?
Hydration changesPolar molecules attract water molecules, mainly through hydrogen bonding. They compete successfully with hydrogen bonds between the water molecules, so they are readily soluble in water.
Is DNA hydrophobic or hydrophilic?
The secret to DNA's helical structureThe environment is therefore hydrophilic, while the DNA molecules' nitrogen bases are hydrophobic, pushing away the surrounding water.
What is DNA insoluble in?
DNA is soluble in water but insoluble in the presence of salt and alcohol. By gently stirring the alcohol layer with a sterile pipette, a precipitate becomes visible and can be spooled out. If there is lots of DNA, you may see a stringy, white precipitate.Is DNA polar or nonpolar molecule?
DNA is a highly polar molecule, which has evolved over millions of years to be stable and functional in aqueous solution.Can you elute DNA in water?
Yes, water can be used to elute DNA from Monarch columns. For maximum elution efficiency, ensure the water is nuclease-free and the pH is between 7-8.5. Milli-Q™ water is often slightly acidic, requiring pH adjustment.Does DNA wash off in water?
DNA quantification results from the human tissue samples reported in ng/μL. Freshwater, swamp water, and saltwater all showed a large loss of DNA over the 72-hour period.What do you think will happen to the DNA sample if water h20 were to be added in the wash buffer 2 step?
Water will help in solubilizing DNA and thus you will lose the isolated DNA .Does DNA form hydrogen bonds with water?
Hydrogen bonding in DNAThese hydrogen bonds between complementary nucleotides are what keeps the two strands of a DNA helix together. Each base can also form hydrogen bonds with the external environment such as with water.